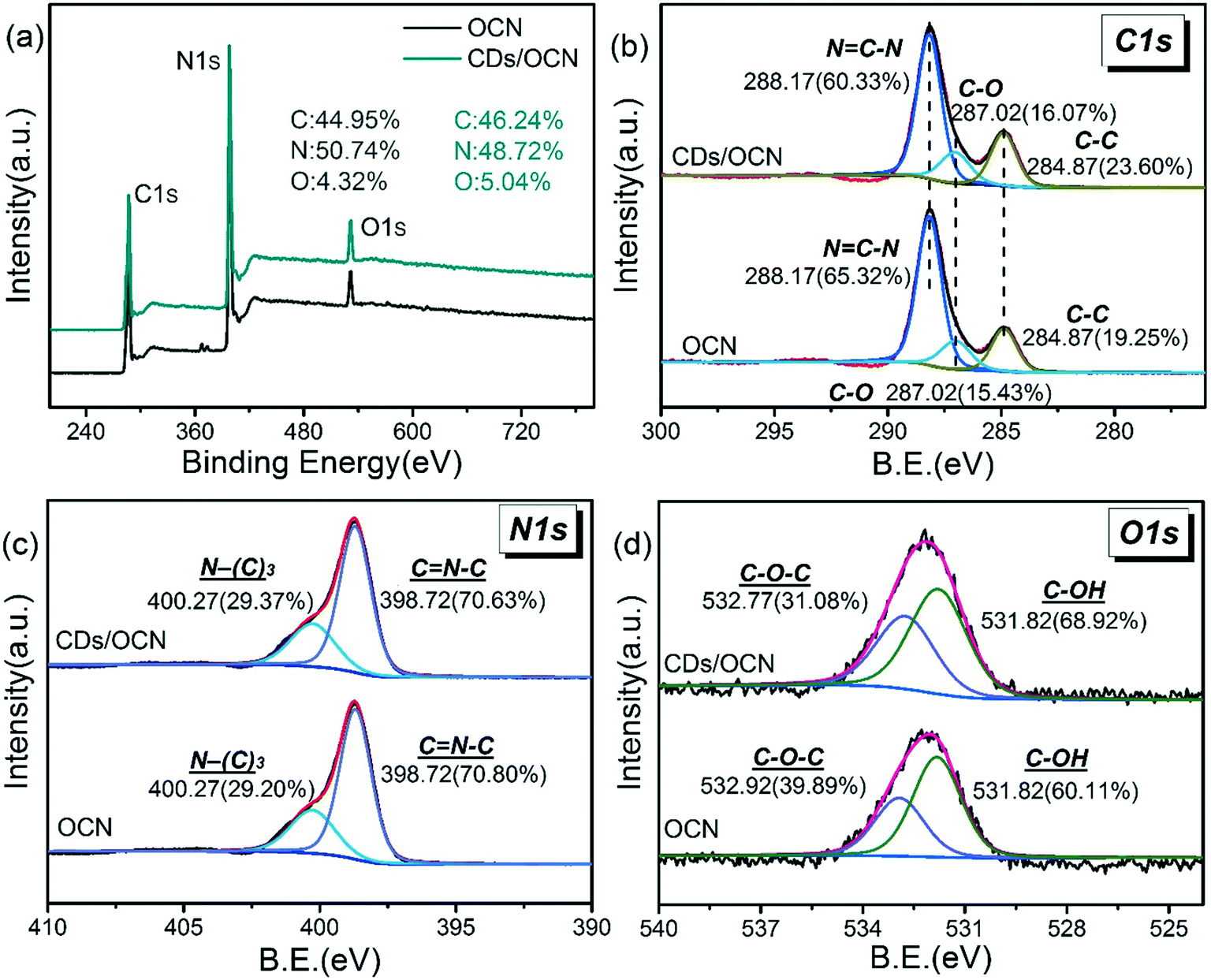
Kubomura, 'Oxidation behavior of mesophase pitch based high modulus carbon fibers' “Achievement in Composites in Japan and the United States” Proc. HIRAI, Preprints for 117 committee of Japan Material Society 15 Nov. of Electron Spectroscopy and Related Phenomena 27 (1982) 39–56. Thomas, “The Chemical Nature of Ion-Bonbarded Carbon: A Photoelectron Spectroscopic Study of 'Cleaned' Surfaces of Diamond and Graphite” Proc. Idem., “Effect of anodic oxidation of coal tar based-carbon fiber on the adhesion in epoxy matrix (part II)-Comparative study of three alkaline solutions” submitted for publication in Composites. Nakanishi, “Anodic oxidation of coal tar based-carbon fiber on the adhesion in epoxy matrix (part I)-Comparison between H2SO4 and NaOH solution-” submitted for publication in Composites. Kubomura, “Oxidation behavior of mesophase pitch based high modulus carbon fibers” Proc. Fujita, Advances in Fiber Composite Materials 12 (1994) 213–236. Due to increased instrument accessibility and reliability, the use of XPS instrumentation has significantly increased around the world. Sherwood, Applied Spectroscopy 44(10) (1990) 1621–1628. The use of peak fitting to extract information from x-ray photoelectron spectroscopy (XPS) data is of growing use and importance. Although the origin of the 2nd graphitic peak is not well understood, it may be related to the amount of oxygen on the GC surface.Ī. For fitting of the high-resolution C1s peak a fixed mixed product of 30 /70. However, the peak shapes of C 1s spectra of anodic oxidized GC after heat treatment at 1500 ☌ in argon atmosphere were almost the same as the C 1s of untreated GC. For chemical investigations X-ray photoelectron spectroscopy (XPS) enables. The ratio of the 2nd graphitic peak area in the C 1s spectrum increased as the electrical charge increased. The 2nd graphitic peak is normally located +0.7–+0.8 eV away from the original C 1s spectrum. However, it was found that the 2nd-carbon peak should also be taken into consideration in the curve fitting process in addition to the effect of asymmetric peak shape of carbon to obtain a consistent value of O 1s/C 1s. Consideration of the asymmetric peak shape of the C 1s spectrum is normally done in order to obtain precise curve fitting results. EDS analysis of cross-sections revealed a gradual P reduction up to 50% towards the inner part of the membrane.An XPS study of anodic oxidized glass-like carbon (GC) was conducted in order to investigate the inconsistency between O 1s/C 1s and oxygen concentration (Ocal.) calculated from the curve fitting results of the C 1s spectrum. The introduction of a neutralization step led to a reduction of P content, which pointed out to the presence of phosphates ionically bound to protonated amines, in addition to phosphate esters. High-resolution XPS spectra regarding C1s, O1s, N1s and P2p are discussed. The phosphate content increased with the reaction time, as shown by XPS and ATR-FT-IR, a P/N atomic ratio of 0.73 being obtained after 48 h of treatment.
Cross-sections were analyzed by SEM fitted with EDS. Surface characterization was performed by XPS, ATR-FT-IR, and SEM. The multiplets were found to t most spectra well, particularly when contributions attributed to surface peaks and shake-up satellites were included. Phosphorylation was carried out at room temperature using the H3PO4/Et3PO4/P2O5/butanol method. Ferrous (Fe2+) and ferric (Fe3+) compounds were investigated by XPS to determine the usefulness of calculated multiplet peaks to t high-resolution iron 2p 3/2 spectra from high-spin compounds. In Figure 3, we see high energy resolution spectra acquired from the copper samples, A. The ability to detect and quantify this shift makes XPS such a powerful analytical technique. Additionally, the negatively charged phosphate functionalities, together with the positively charged amine groups from chitosan, are expected to provide chitosan with an amphoteric character, which may be useful as a combinatorial therapeutic strategy, by simultaneously allowing the immobilization of signalling molecules like growth factors. peaks in finer detail, then we see that even for the same element, the peak energy may shift depending on the surface chemistry. Phosphate groups chelate calcium ions, thus inducing the deposition of an apatite-like layer known to improve the osteoconduction of polymer-based implants.

Phosphorylation may be of particular interest in materials for orthopaedic applications, due to the cation-exchange properties of phosphate functionalities. The technique was first known by the acronym ESCA (Electron Spectroscopy for Chemical Analysis). In the present work, the surface of chitosan membranes was modified using a phosphorylation method carried out at room temperature. X-ray Photoelectron Spectroscopy (XPS) was developed in the mid-1960s by Kai Siegbahn and his research group at the University of Uppsala, Sweden.
